ELPSS delivers end-to-end scientific and quality solutions that transform scientific analytical data into process intelligence and regulatory confidence.
We provide scientific leadership, regulatory strategy, and analytical oversight, leveraging a vetted network of ISO-certified laboratories to execute testing when needed
What We Do
Analytical Strategy & Oversight
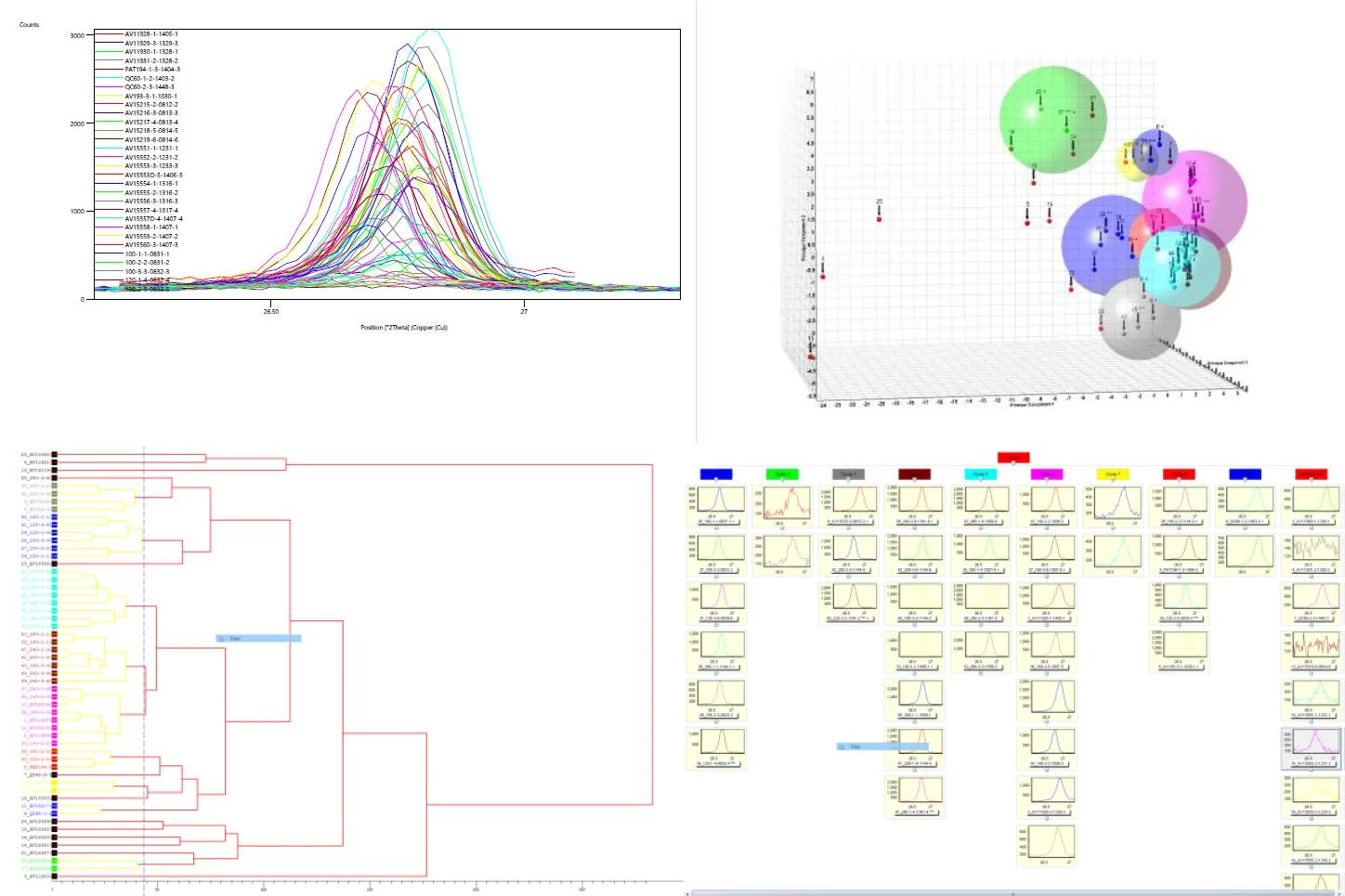
Material Characterization (e.g., Organics, Liquids, Metals, Nonmentals, Hydrocarbons, Glycols) through tandem techniques (e.g., ICP-OES, ICP-MS, XRD, LC-MS/MS, GC-MS/MS)
Executed through vetted ISO-certified laboratory partners
Quality control processes (e.g., stability studies, 21 CFR 211, 111, 117 processes)
manufacturing/production processes improvements through data analytics
Why Choose Us
Independent scientific judgment
No lab conflict of interest
Risk-based regulatory approach
Deep FDA / ISO / GMP fluency
Single accountable scientific lead
Defensible data packages—not just reports
Focus on
Corrosion
Material Characterization, waste control
Nutraceutical & Pharmaceutical
21 CFR 111, 211, and 117 interpretations
ICH implementations
Supplier qualification strategy
Contaminant risk assessment
Stability and shelf-life defensibility
Method lifecycle strategy
Validation oversight
Regulatory alignment
Manufacturer and lab audits
Mining
Material characterization
Bioleaching strategy